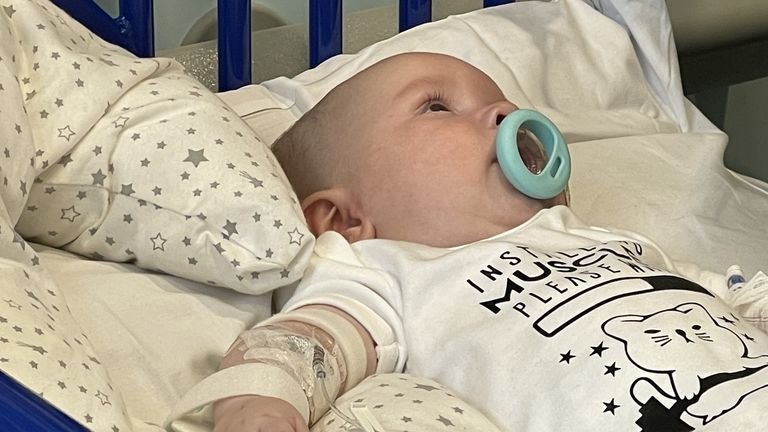
A five-month-old baby with spine disease has become the first NHS patient in England to be treated with a potentially life-saving drug that can prolong life.
Arthur Morgan was diagnosed with spinal muscular atrophy (SMA) earlier this month.
He received the unique American gene therapy, Zolgensma, at Evelina Children’s Hospital in London on May 25.
The treatment has been called the most expensive drug in the world – with a list price of £ 1.79million per dose.
It was made available on the NHS after the health service struck a deal with manufacturers Novartis Gene Therapies in March.
Baby Arthur, born six weeks prematurely in December, underwent a gene therapy infusion last week after being diagnosed with SMA less than three weeks earlier.
Until two years ago, no treatment options were available for children with this disease.
His father, Reece Morgan, who works as a freelance plasterer, said: “When we heard that Arthur would receive the treatment and be the first patient, I broke down.
“It had been such a whirlwind of a few weeks, filled with a lot of anxiety and adaptation, as we learned of his condition and what it might mean for him and our family.”
The 31-year-old added: “We still don’t know what the future holds, but it gives Arthur the best possible chance to give him the best possible future.”
Babies born with type 1 ADS – the most common form of the disease – suffer from progressive muscle weakness, loss of movement, difficulty breathing, and have a life expectancy of only two years.
But studies have shown that just one treatment with Zolgensma can help babies with Zolgensma sit, crawl and walk, and also prevent them from having to be put on a ventilator.
Four specialist NHS centers across England can now administer the …
More information about this article Read More
Source: news.sky.com
This notice was published: 2021-06-01 00:14:00